 The number of "octet electrons" is equal to the number of valence electrons that each atom will have when they have the same electron configuration as the nearest noble gas (the octet rule). You won't find the term "octet electron count" in any textbook because, as far as I know, I made it up. For example, NH 4 + will have one fewer valence electron that the nitrogen and four hydrogens would have because it has a +1 charge.Ĭount the total octet electron count in the molecule. To do so, subtract the ionic charge from the valence electron count. Occasionally, you'll have to find the Lewis structure for a polyatomic ion. Therefore, the number of valence electrons for this molecule is 4 + (4 × 7) = 32. The single carbon atom contains four valence electrons, and each of the four chlorine atoms contains seven valence electrons. Elements in Period Three, Four, etc (on the periodic table) can hold more than 8 valence electrons.Lewis structures (named for chemical theorist Gilbert Newton Lewis) are pictures that show all of the valence electrons and atoms in a covalently bonded molecule.Ĭount the total number of valence electrons in the molecule.Īs an example, let's use carbon tetrachloride, CCl4.S and P sometimes have more than 8 val. 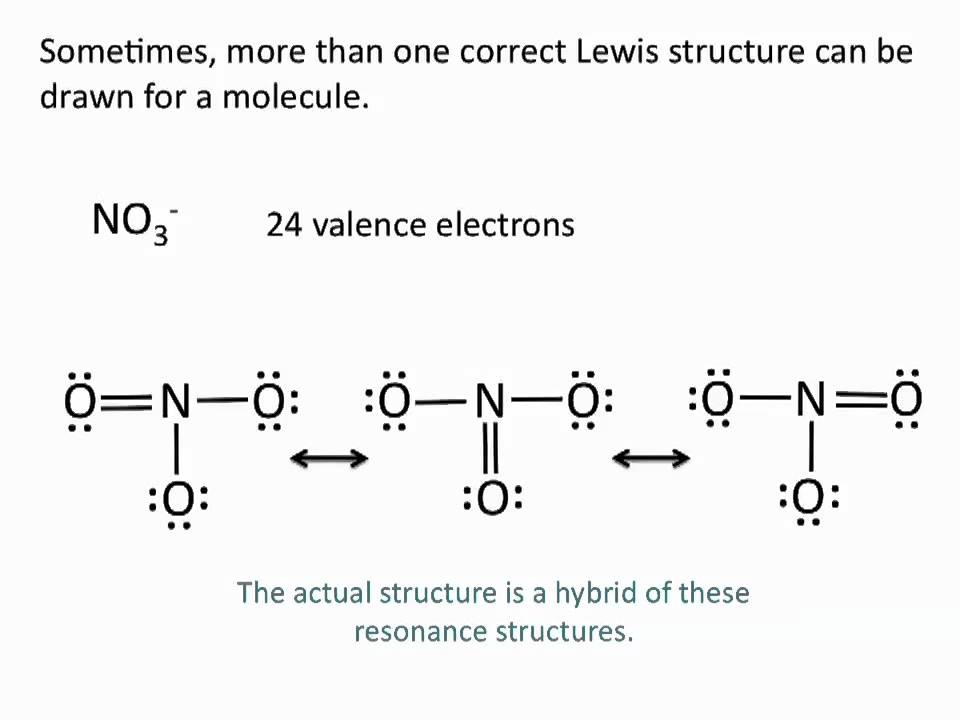
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
